The Dolomite mineral is commonly found in deposits of a sedimentary rock called dolostone, was named after the French mineralogist Deodat de Dolomieu. Basically, there are two types of materials referred to as dolomite: a true chemically uniform calcium magnesium carbonate with the chemical formula CaMg(CO3)2, and a dolomitic limestone an irregular mixture of calcium and magnesium carbonates.
It is believed that dolomite was formed by replacement of some of the calcium in a calcium carbonate limestone deposit with magnesium while the sediment was undergoing lithification, being transformed from layers of dead clam and other sea animal shells into crystalized calcite or calcium carbonate. The resulting dolomite mineral, CaMg(CO3)2 is a real double salt.
Salient features of our Natural Dolomite
Higher degree of purity, wet ability and whiteness
Our natural dolomite is popular for its shear and compressive strength
Fire resistive and solid
Long lasting life and stiffness is the strength of our mineral dolomite
High Specific Gravity and High Bulk Density mineral with Less Reactive
Burnt/calcined Dolomite is used for Refractory
Dolomite is widely used in Iron Steel Industries due to its strength and compatibility in process of purifying Iron Steel
Used as a substitute of lime for construction and building products
White Dolomite powder as a filler for asphalt and concrete, paints, plastics, rubber, soaps and detergent applications
For adhesive formulation
Dolomite is also used in some cement, pharmaceutical, fodder, Glass and ceramic industry as a source of magnesium
As a sintering agent in iron ore pelletization or as a flux agents in steel making
In agriculture, dolomite uses serves as pH control, while the chemical industry uses the mineral in making magnesium salts
Note: Specifications and chemical analysis can be furnished only when we get to know the grades/applications desired by you.
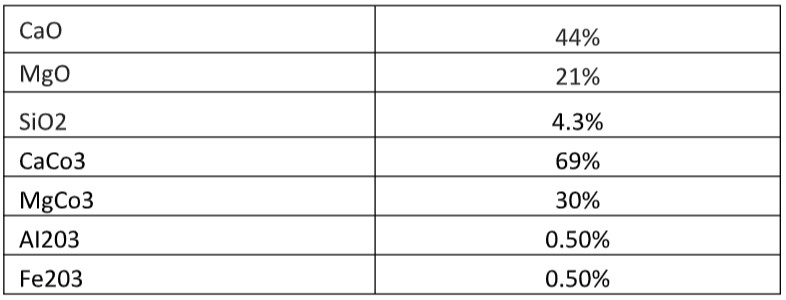
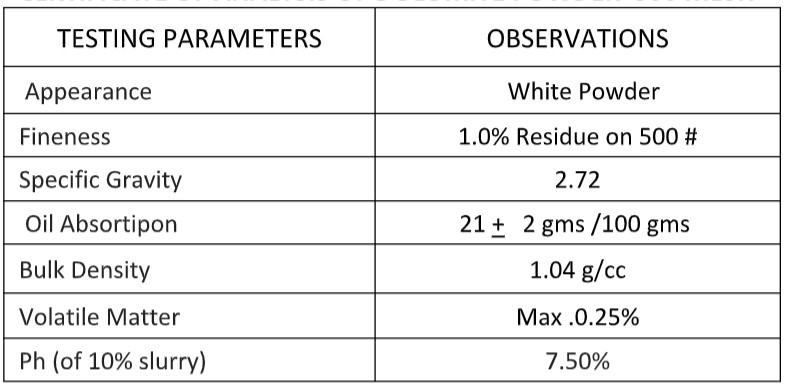
Certificate of Analysis of Dolomite Powder 240 Mesh
Chemical Analysis